Difference Between Fractional and Simple Distillation
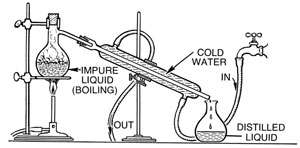
In chemistry, we are taught how to separate mixtures, and one of the most interesting ways to separate mixtures is through distillation. Distillation is a common method that is often used in separating liquid mixtures, based on the differences in the volatilities of substances. Distillation is a physical process, as it does not rely on any chemical reaction.
Distillation is an essential process for many industries – from power generation to beverage production.
There are two common processes in distillation, the simple distillation and the fractional distillation. They are very similar in many ways. They both try to separate mixtures, and use the same equipment. The principles are also essentially the same. However, distinctively, fractional distillation will need additional equipment, and that equipment is called the ‘fractionating column’.
A fractionating column is used in fractional distillation, because the liquid mixtures involved in the process, have boiling points that are similar to each other. The fractionating column acts as a minor obstruction to the rising gas. It will prevent the ‘not so’ pure vapor from passing through. The gas will condense on the surface area of the packing material in the fractionating column, and will be reheated by the rising hot gas, to be vaporized again until it becomes ‘pure’.
Repeated distillation processes take place, and this is also referred as rectification. Each distillation (vaporization-condensation) cycle is called theoretical plate. More cycles would mean better separations, and lead to purer results. This is why the process is called fractional distillation, because it is basically composed of two or more cycles of distillation.
When the substances in a mixture have significant differences in boiling points, a simple distillation is adequate to be implemented. As the name suggests, it is as simple as it gets. No fractionating column is required for use. A single distillation cycle is enough to separate the mixture. When one has to separate a liquid from a solid, the simple distillation is usually the method of choice since, obviously, solid and liquid phases have very differing volatilities.
Summary:
1. Simple distillation is the method used to separate substances in mixtures with significantly different boiling points, while fractional distillation is used for mixtures containing chemicals with boiling points close to each other.
2. Simple distillation will only have one distillation (vaporization-condensation) cycle in the whole process, while the fractional cycle will have at least two cycles.
3. Fractional distillation will use additional equipment called the ‘fractionating column’, while the simple distillation will not need that equipment.
4. Simple distillation is often used to separate the liquid substance from the solid substance. Doing such separation with fractional distillation is not sensible.
- Difference Between Lymphoma and Leukemia - March 7, 2024
- Difference Between CP and CPK - June 8, 2010
- Difference Between PPTP and L2TP - June 7, 2010

That’s awesome yaar♥♥
we actually asked for the differences not theory part to explain and for differences there should be a tabular column or a line between that helps the readers to understand easily this is not a command instead a suggestion from my side and everybody’s side by the way that theory was really nice
yes, the explanation really nice, and understandable but you should draw this in a tabular form or put in line between them so that it we easily recognized, nice work
Sure
who wrote this
whaj is most sensible technique to separate palm oil and kerosene?please
Nice one I love the summary
The explanation is very heplfull to me and it was wrote in very simple language that anybody can understand without doing any hard work
Good but it should be in tabular form
Yaa.. It’s good I got many things in this but I want differences between this there should be is in tabular form. It should be in like a column
Book
Summary is nice but there should be tabular column between them so it can be more easy to understand….but really helpful to me…
This is good website
Hii
I am anuj
Nice answer Yar…^_^✌️ but why in differences question u wrote summary..??
This is good but “PUT IN A TABULAR FORM”?!!
“PUT IN A TABULAR FORM”. It is good but go straight to the point.