Difference Between Density and Volume
Density vs. Volume
Density and volume are two scientific concepts that pertain to the physical properties and characteristics of matter. These properties often describe the quality or property of a given object. Both concepts are usually encountered in the field of physics and both act as measurement tools for three-dimensional objects. Both properties can be applied for three stages or states of matter, which are: solid, liquid, and gas.
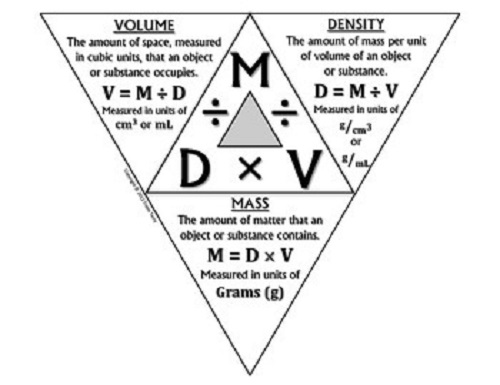
“Density” is defined as the mass per unit of volume. To put it simply, it tackles the concept of how much matter or mass is inside an object in the space that it occupies. It also describes the relationship between an object’s mass and volume.
Density is symbolized by the letter “D” and is a scalar quantity of matter. The formula is p = M/V, or density equals mass divided by volume. This formula is only applicable for objects with uniform compositions, or solid objects. From this same formula, with a few adjustments, the formula for volume and mass can also be derived. In experiments, mass is often determined first before the volume.
Density is measured in the following units: pounds per cubic foot, grams per cubic centimeter, and kilograms per cubic centimeter.
Different substances have different densities. Due to this fact, this property of matter can be used to determine whether a certain sample or substance is genuine. Density can change or vary when temperature or pressure is applied.
Volume, in comparison, is a component of density. As a property, volume is concerned with how much space a particular object occupies. It is represented by the letter “V.” In finding density, volume is one of the two key components, the other one being mass.
In a three-dimensional object, the shape’s volume can be determined by multiplying the dimensions, which comprise of the object’s length, width, and height. Specific shapes like a cube, cylinder, prism, rectangular prism, pyramid, cone, circle, sphere, and other shapes have specific formulas for determining their respective volume.
For liquids and gases, the sample substances are put into a container and calculated. Volume is expressed in units and subunits like cubic meters for solids and cubic liters for liquids and gases. Density and volume have an inverse relationship with each other. If density increases, the effect will be a decrease in volume. In contrast, if the volume increases, density decreases.
Summary:
1.Both density and volume are physical properties of matter. They are present in the traditional stages of matter, which are solid, liquid, and gas. Both density and volume have a specific formula regarding solids or regular-shaped objects. In measuring liquids and gases, there is a little deviation from the traditional approach or formula.
2.Density is represented by the letter “D,” while volume is recognized using the letter “V.”
3.Density measures the amount of matter present in an object. Meanwhile, volume is concerned with the amount of space that an object occupies.
4.The density formula for solid or three-dimensional objects comprises two components – mass and volume. In this view, volume is a component of density. On the other hand, the volume of a regular shape is determined by three dimensions: length, width, and height. In many instances, volume is measured by the use of a graduated cylinder, water, and a specific object.
5.In terms of units, density units are a composite and involve the mass and volume components. In contrast, there is only one component involved in the volume, which is the unit used for volume only.
6.Density and volume have inverse relationships in accordance with the mathematical formula for density.
7.Density and volume are very important concepts to consider especially in experiments. Determining these properties is very helpful when combining or working with substances.
- Differences Between Fraternity And Sorority - January 8, 2014
- Differences Between Lucite and Plastic - January 7, 2014
- Differences Between Oil and Butter - January 6, 2014
Search DifferenceBetween.net :
 Email This Post
: If you like this article or our site. Please spread the word. Share it with your friends/family.
Email This Post
: If you like this article or our site. Please spread the word. Share it with your friends/family.
2 Comments
Leave a Response
References :
[0]http://www.hlswatch.com/2015/05/28/exploring-a-possible-strategic-analogy-density-massvolume/


How can one get confused with density and volume. Would anyone get confused about the difference between time and speed??
the letter describing the density is not the p, but the Greek lowercase {rho}. Looks similar, but there is also a difference between 0 (number zero) and O (letter O).
how do i use the sea salt for my tragus piercing?