Difference Between Chiral and Achiral
A chiral is an object that is not identically superimposable with a mirror image of itself. An achiral is an object that is identically superimposable with a mirror image of itself.
What is Chiral?
Definition of Chiral:
Chiral is a term that is used to describe an object or item that is not completely and identically superimposable with a mirror object of itself. Chirality is a feature that is often used in chemistry to describe the configuration of atoms of molecular structures.
Symmetry of Chiral:
Any object that is chiral then is not symmetrical since symmetry occurs when objects do have identical mirror images along a plane. Such objects that are asymmetrical but have the same composition in chemistry are known as stereoisomers. There are different types of stereoisomers that occur in chemistry, which can be very significant in terms of the reactivity of a substance. Although chiral objects will not have reflective symmetry they may still have rotational symmetry, which means that they look the same when rotated 360 degrees.
Examples of Chiral:
Everyday chiral objects are, for instance, scissors or golf clubs, where you can choose left-handed or right-handed. In chemistry, chirality can be used to describe certain molecular configurations. A tetrahedral carbon that is bonded to different atoms is most often chiral with non-identical mirror images possible if the carbon is asymmetrically bonded to other atoms. These mirror images in chemistry are referred to as enantiomers and they also have the property of being either right-handed or left-handed in a sense, but in chemistry, this handedness is designated as R or S.
Significance:
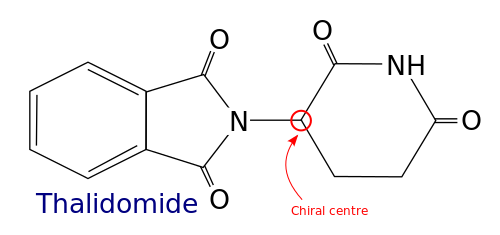
Chirality is an important aspect that chemical engineers must understand, and they must comprehend the significance of stereoisomerism and enantiomers on chemical function. If a drug is designed that is not of the correct form, in terms of chirality, this can have drastic consequences for patients, and may even lead to further illness or death. Researchers investigating amyloid proteins that are implicated in causing brain disease have discovered chirality in the structure of the amyloids, which may be useful in developing treatments.
What is Achiral?
Definition of Achiral:
Achiral is a term used to describe an object that is completely and identically superimposable with a mirror object of itself. The nature of chirality is that generally, only simple molecules such as hydrocarbons are likely to be achiral.
Symmetry:
Objects and molecules that are achiral have a reflective center of symmetry, and they thus appear identical even when viewed in a mirror. Such objects that are achiral also have what is called rotational symmetry.
Examples of Achiral:
An example of an everyday item that is achiral is a hardware nail. A mirror image of such a nail is identical to the actual object, and it has symmetry around a plane. In chemistry, the molecular structure of propane is chiral since the mirror image is identical to the original molecular object.
Significance:
Achiral objects do not show stereoisomerism and thus there is no concern when chemical engineers design such molecular structures since the molecules can only bond in one particular way.
Difference between Chiral and Achiral?
-
Definition
A chiral object is not completely and identically superimposable with a mirror object of itself. In comparison, an achiral object is completely and identically superimposable with a mirror object of itself.
-
Reflective symmetry:
Chiral objects do not have reflective symmetry since they are asymmetrical around a line of division. Achiral objects do have reflective symmetry because they are symmetrical around a line of division.
-
Left-handed and right-handed forms
An object that is chiral has both left-handed and right-handed forms, known as S and R for the configuration of molecular objects. An object that is achiral does not have right or left-handed forms, or S and R molecular forms.
-
Identical mirror images
Chiral objects and molecules never have identical mirror images since they are asymmetrical. Achiral objects always have identical mirror images since they are symmetrical.
-
Enantiomers
Chiral molecules often have enantiomers since stereoisomerism is present, while achiral objects do not have enantiomers.
-
Examples from everyday objects
Some everyday objects and items that show the chiral form include scissors and golf clubs, where a person can buy right-handed or left-handed forms. Some everyday items that have an achiral form are hardware nails.
-
Examples from chemistry
A chemistry example of a chiral molecule is a molecule that has a tetrahedral carbon present to which different atoms bond to produce an asymmetrical structure. A chemistry example of an achiral molecule is the propane molecular structure.
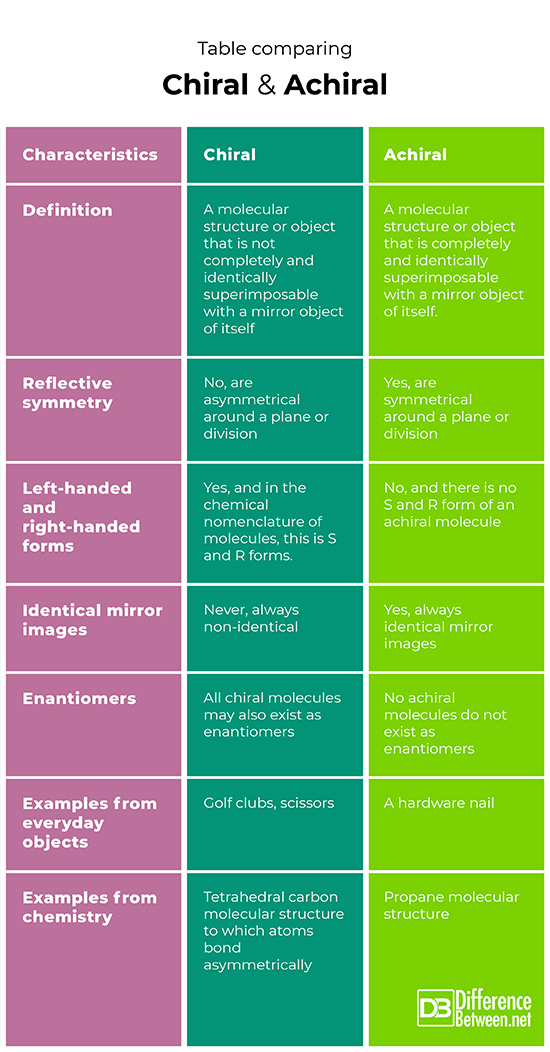
Table comparing Chiral and Achiral
Summary of Chiral Vs. Achiral
- Chirality is more commonly observed in complex molecules than is achirality.
- Achiral objects show an identical mirror image to the actual object.
- Chiral objects do not show an identical mirror image to the actual object because of the asymmetrical configuration of the object or item.
- Both achiral and chiral objects can show rotational symmetry around 360 degrees, but chiral objects do not have reflective symmetry.
- Chirality is an important aspect of chemistry since different enantiomers may have different reactivities.
- Knowledge of the chirality of molecular structures can help in the treatment of diseases and in the design of medication.
- Difference Between Rumination and Regurgitation - June 13, 2024
- Difference Between Pyelectasis and Hydronephrosis - June 4, 2024
- Difference Between Cellulitis and Erysipelas - June 1, 2024
Search DifferenceBetween.net :
Leave a Response
References :
[0]Reusch, William. “Stereoisomers II: Chirality and Symmmetry.” Chemistry. Michigan State University, 2008, https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/chapt11.htm
[1]Rubin, Noa, et al. "Chirality of amyloid suprastructures." Journal of the American Chemical Society 130.14 (2008): 4602-4603.
[2]Image credit: https://commons.wikimedia.org/wiki/File:Soai_reaction_scheme.png
[3]Image credit: https://upload.wikimedia.org/wikipedia/commons/thumb/2/22/Thalidomide_molecule_with_chiral_carbon_annotation.svg/500px-Thalidomide_molecule_with_chiral_carbon_annotation.svg.png