Difference Between Acetone and Xylene
Acetone is a chemical that is a form of a ketone which has formula CH3COCH3. Xylene is a chemical that is a type of hydrocarbon that has formula C6H4(CH3)2.
What is Acetone?
Definition:
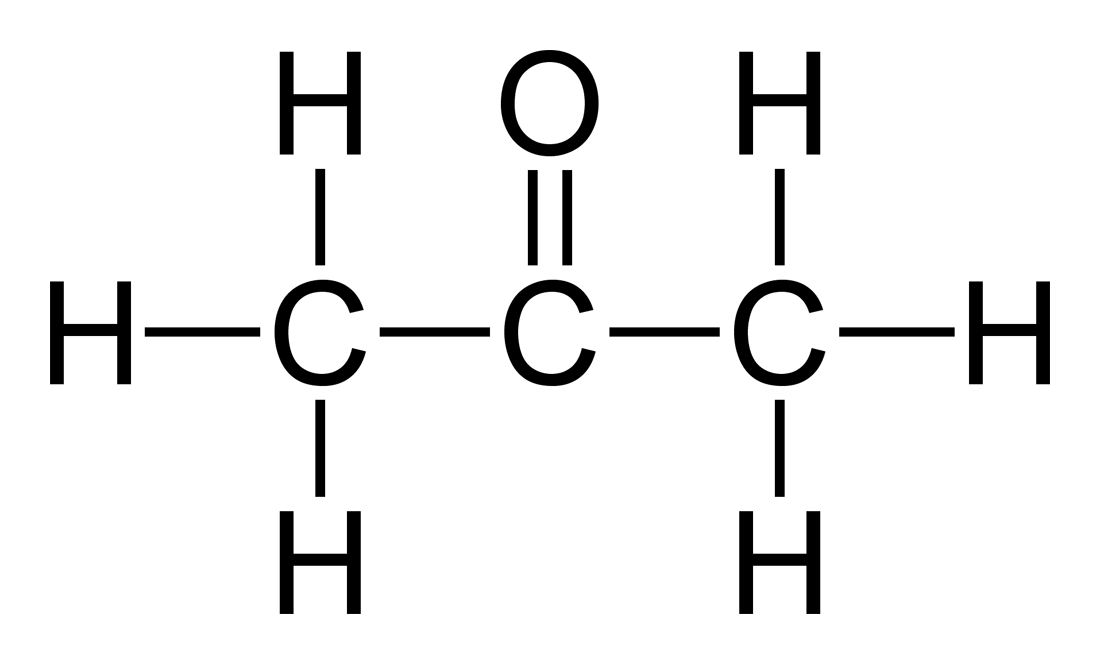
Acetone is considered to be one of the ketone molecules which has the formula of C3H6O or
CH3COCH3.
Properties:
Acetone is commonly present as a colorless liquid substance which also has a molecule weight of about 58.08 g/mol. The smell of acetone is quite distinctive to most people since it smells a lot like fruit. It is also a flammable substance that needs to be handled with care as a result.
Formation:
The cumene method that involves hydroperoxide is how acetone is synthesized in industry. Acetone is a byproduct of the metabolism of lipids in the animal body, and thus can be formed in humans, but is particularly noticeable in people who have uncontrolled diabetes.
Uses:
The most common use of acetone is as a solvent to remove nail polish from fingernails, and as a way to remove ink or paint from surfaces. It is occasionally also used in industry to manufacture fibers and has some bacteriostatic abilities when used against certain species of bacteria.
Safety:
In small amounts, the acetone is not very harmful but it does produce vapors that can irritate the mucous membranes of the eyes and the cells of the skin. It is also a flammable chemical and so people should be careful to not use too much in an enclosed space or near an ignition source.
What is Xylene?
Definition:
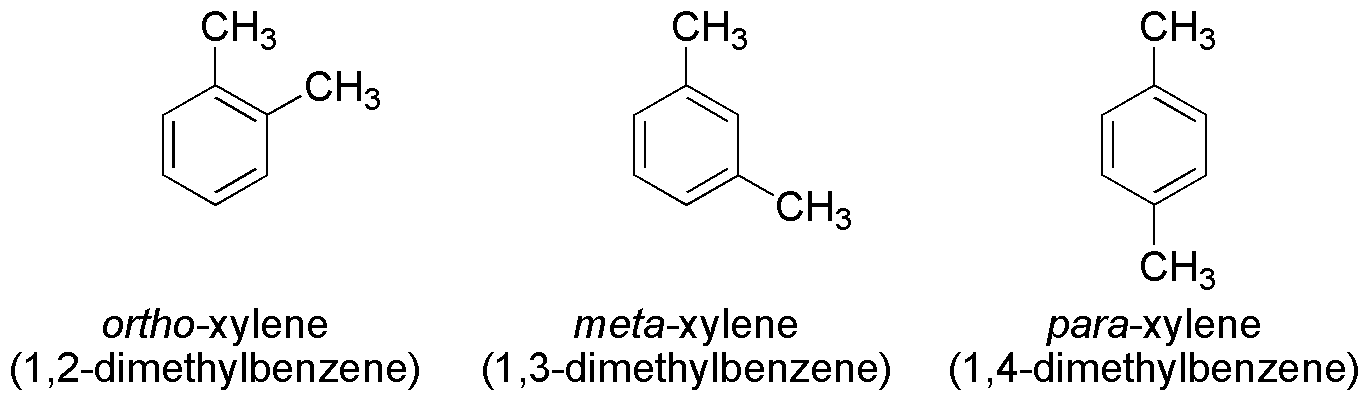
Xylene is a chemical that is a type of hydrocarbon molecule which is often present in one of three forms (isomers) which contain a benzene ring. These isomers differ only in where the methyl group is attached to the molecule, and they all have the formula C6H4(CH3)2.
Properties:
The xylene can occur as a gas or a liquid that is colorless and has a distinctive smell that has been described as sweet. The molecular weight of xylene is 106.168 g/mol. It is also a flammable substance that therefore needs to be used with care. It is not soluble in water and it is quite toxic to people.
Formation:
Xylene can be formed in a reaction that involves benzene and toluene. The chemical reaction is an alkylation type that results in the production of the xylene.
Uses:
Xylene is used as a solvent for lacquers and paints. It is also used in the leather industry and is sometimes part of the process when plastics are manufactured. Xylene is also used in the staining of tissues when making microscope slides.
Safety:
Xylene is a potentially dangerous chemical since it is flammable and has been shown to cause irritation of the mucous membranes and skin. It has also been shown to be toxic to the body if inhaled and thus individuals working with xylene should take precautions to avoid contact with skin or with breathing in the fumes. Xylene can be harmful to the nervous system and cause damage to organs like the lungs, kidneys, and liver.
Difference between Acetone vs. Xylene?
-
Definition
The acetone molecule is a type of chemical which has the formula C3H6O or
CH3COCH3. The xylene molecule is a type of chemical which has the formula C6H4(CH3)2.
-
Molecular weight
Acetone has is a smaller molecule with the molecular weight of 58.08 g/mol. Xylene is a larger molecule with the molecular weight of 106.168 g/mol.
-
Type of molecule
The acetone molecule is a type of ketone. The xylene molecule is a type of hydrocarbon
-
Number of oxygen atoms
The acetone has one oxygen atom in the molecular structure. The xylene has no oxygen atoms anywhere in the molecular structure.
-
Formation in industry
A cumene hydroperoxide method is used to make acetone synthetically. An alkylation is done to attach methyl groups to benzene to make xylene and is often made from petroleum.
-
Formation in animals
Acetone can be formed in animals when lipid molecules are catabolized and ketone bodies are produced. Xylene is not a substance that is normally formed in animals or other living organisms.
-
Odor
The odor of acetone is similar to a fruit. The odor of xylene is that of a sweet substance.
-
Uses
Acetone can be used to take off nail polish, paints or even lacquers. Xylene can be used for making histological preparations, and certain plastics. It can also be used as a solvent and cleaning agent.
Table Comparing Acetone vs. Xylene
Summary of Acetone vs. Xylene
- Acetone and xylene are both flammable substances that have a distinctive odor.
- Acetone and xylene also both work well as solvents to remove lacquers and paints.
- Acetone is a smaller molecule than xylene and it contains an oxygen atom in the molecular structure.
- Xylene has a sweet smell and is toxic to humans.
- Both xylene and acetone can irritate the mucous membranes so people should be careful when using these chemicals.
- The smell of acetone is more like that of a fruit, and it can also be harmful to humans.
- Acetone is a chemical that can be formed in the body when fats are broken down.
- Difference Between Rumination and Regurgitation - June 13, 2024
- Difference Between Pyelectasis and Hydronephrosis - June 4, 2024
- Difference Between Cellulitis and Erysipelas - June 1, 2024
Search DifferenceBetween.net :
1 Comment
Leave a Response
References :
[0]Kandyala, Reena, Sumanth Phani C. Raghavendra, and Saraswathi T. Rajasekharan. "Xylene: An overview of its health hazards and preventive measures." Journal of oral and maxillofacial pathology: JOMFP 14.1 (2010): 1.
[1]Image credit: https://commons.wikimedia.org/wiki/File:Acetone-2D-flat.png
[2]Image credit: https://commons.wikimedia.org/wiki/File:Xylenes.png


Informative from an academic.standpoint, but not as informative feom an aoplication and usage standpoint. In what circumstances would employing acetone be the better choice, and vice versa. I am thinking primarily as a solvent and.cleaner of paints, greases and oils. Also, how do these compare to Toulene and posibly nitro-methane when seeking the strongest solvent. I know n.m. is excellent fro breaking down substances such as crazy glue…Thanks for your thoughts…Alex