Difference Between Retinol and Retinyl Palmitate
Retinol is a chemical known as vitamin A that is very abundant in the body. Retinyl palmitate is a chemical that is made from retinol and palmitate and is known as vitamin A palmitate.
What is Retinol?
Definition:
Retinol is a substance that is also known as vitamin A and is one of the most abundant forms in which this particular vitamin is found in the human body. The amount of retinol in the blood plasma of many different species of mammals is about 1–2 μM.
Structure:
Retinol is a molecule that is classified as being an alcohol and it has the molecular formula of C20H30 and molecular mass of 286,45 g/mol. It is a fat-soluble vitamin which the liver does store. The molecule has double bonds and is quite easily degraded by oxidizing factors such as acidic substances and also heat. The boiling point of retinol is about 137 degrees Celsius and the melting point is about 63 degrees Celsius.
Functions:
Vitamin A is an important nutrient that needs to be taken into the human body through the diet. Too little or too much vitamin A can be very harmful. Retinol is an important molecule that is converted into retinal which is a molecule that is needed and used by the photoreceptor cells of the eyes, which are responsible for vision. Too little vitamin A can cause deficiency symptoms and too much retinol can cause problems since it is stored in the liver.
Formation:
Retinol is formed from provitamin A in the body from molecules taken in through the diet or through supplements. The enzyme β-carotene 15, 15′-dioxygenase has been shown to make vitamin A from carotenoid pigments such as carotene that are taken into the body. Thus, vitamin A can be made from food that contains carotenoids.
What is Retinyl Palmitate?
Definition:
This is an ester found in the liver of animals, which is also known as vitamin A palmitate. It is made from a chemical reaction of retinol and palmitate.
Structure:
The molecular formula for retinyl palmitate is C36H602 and the molecular mass of the chemical is 524,86 g/mol, and the melting point is about 28.5 degrees Celsius. The ester of retinol has better stability than retinol and is why this is often the form that vitamin A occurs in when present in supplements or cosmetic products. Even though the retinyl palmitate is a different molecular form than retinol it is also taken up through the intestine in the same manner and can be stored in the liver of the body.
Functions:
The retinyl palmitate is usually the form in which vitamin A supplements are made available since it is more stable than conventional retinol, which means that it is easier to use and lasts longer when made into pills or tablets that people buy to take as a supplement. It can be taken as pills or capsules or even injected into the body. It is often used as a supplement for the treatment of vitamin A deficiency in people.
Formation:
Retinyl palmitate is one of the esters that can be formed from vitamin A. It is produced by an esterification reaction. The hydroxy group of the retinol undergoes a chemical reaction with the carboxy group of palmitic acid to form the retinyl palmitate.
Difference between Retinol and Retinyl Palmitate
Definition
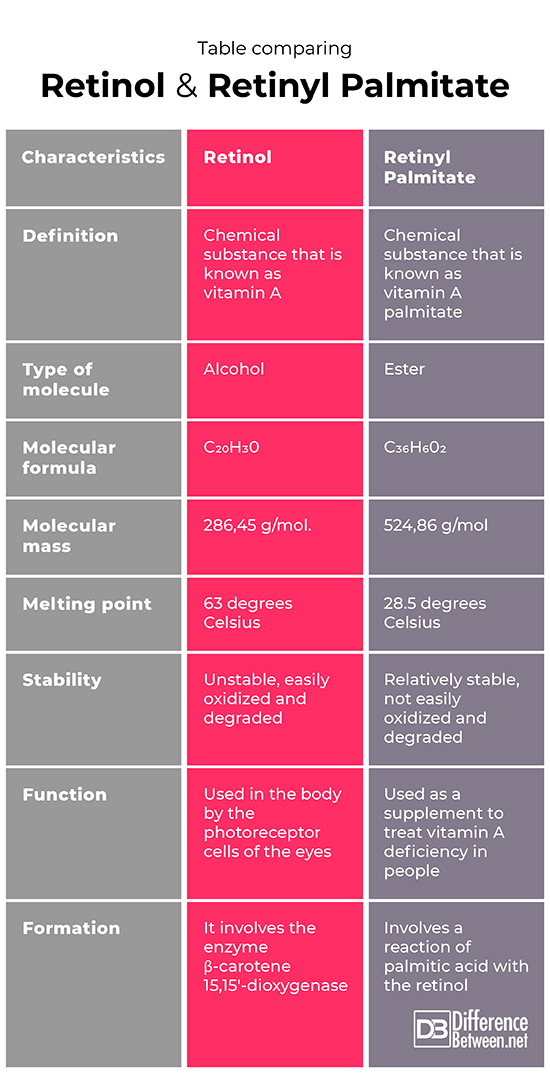
Retinol is a chemical substance that is also known as vitamin A. Retinyl palmitate is a chemical substance that is called vitamin A palmitate.
Type of molecule
The retinol is a type of molecule that is classified as an alcohol. The retinyl palmitate is a molecule that is classified as an ester.
Molecular formula
The molecular formula of the retinol molecule is C20H30. The molecular formula of the retinyl palmitate molecule is C36H602.
Molecular mass
The molecular mass of the retinol molecule is 286, 45 g/mol. The molecular mass of the retinyl palmitate molecule is 524, 86 g/mol.
Melting point
The retinol molecule has a melting point of 63 degrees Celsius. The retinyl palmitate molecule has a melting point of 28.5 degrees Celsius.
Stability
Retinol is an unstable molecule that is easily broken down and oxidized. Retinyl palmitate, by comparison, is a stable molecule that is not as easily degraded or oxidized.
Function
The vitamin A, retinol is used in the body by photoreceptor cells in the eye and is therefore important in vision. The retinyl palmitate (vitamin A palmitate) is often the form found in supplements used to treat people who have a lack of vitamin A in their bodies.
Formation
The formation of retinol involves the action of the enzyme involves the enzyme β-Carotene 15, 15′-dioxygenase. The formation of retinyl palmitate involves a reaction of palmitic acid with retinol.
Table comparing Retinol and Retinyl Palmitate
Summary of Retinol Vs. Retinyl Palmitate
- Retinol and retinyl palmitate are both forms of vitamin A that can be used in the human body.
- Retinol is an alcohol that is less stable than retinyl palmitate and relatively easy to oxidize as a result.
- Retinyl palmitate is often the form of vitamin A that is made and used in supplements to help people who are deficient in vitamin A, and it is often called vitamin A palmitate.
- Retinyl palmitate is made from a reaction between palmitate and retinol.
- Difference Between Rumination and Regurgitation - June 13, 2024
- Difference Between Pyelectasis and Hydronephrosis - June 4, 2024
- Difference Between Cellulitis and Erysipelas - June 1, 2024
Search DifferenceBetween.net :
1 Comment
Leave a Response
References :
[0]O'Byrne, Sheila M., and William S. Blaner. "Retinol and retinyl esters: Biochemistry and physiology Thematic Review Series: Fat-soluble vitamins: vitamin A." Journal of lipid research 54.7 (2013): 1731-1743.
[1]von Lintig, Johannes, and Adrian Wyss. "Molecular analysis of vitamin A formation: cloning and characterization of β-carotene 15, 15′-dioxygenases." Archives of biochemistry and biophysics 385.1 (2001): 47-52.
[2]Yokota, Satoshi, and Shigeru Oshio. "A simple and robust quantitative analysis of retinol and retinyl palmitate using a liquid chromatographic isocratic method." Journal of food and drug analysis 26.2 (2018): 504-511.
[3]Image credit: https://commons.wikimedia.org/wiki/File:Retinol_3D_spacefill.png
[4]Image credit: https://en.wikipedia.org/wiki/File:Retinyl_palmitate_spacefill.png

Good Evening.
On the very outset. I like to thank you for such a good article.
My question:
How these two molecules help in the treatment of Retinopathy in (1)acute stage (2) chronic stage (3) primary stage.