Difference Between Affinity and Avidity
While theoretical thermodynamics of antigen-antibody interactions are well known, their practical use in detection or determination of autoantibodies is scarce. For functional antibodies, determination of the optimal affinity allows achievement of the maximum therapeutic benefit. Antibody affinity has been studied for almost half a century and since the inception of affinity chromatography three decades ago, it has developed into a powerful tool. Affinity describes the interaction in an individual binding site. Knowledge of the affinity as well as construct and antigen properties is essential for evaluating the link between affinity and potency for a functional antibody. However, in addition to affinity, there is yet another binding concept called avidity, which is the overall binding strength of the interaction between a multivalent antigen and a multivalent antibody. Despite the clear differences between the two, the terms affinity and avidity are often indiscriminately used. Let’s take a look the two.
What is Affinity?
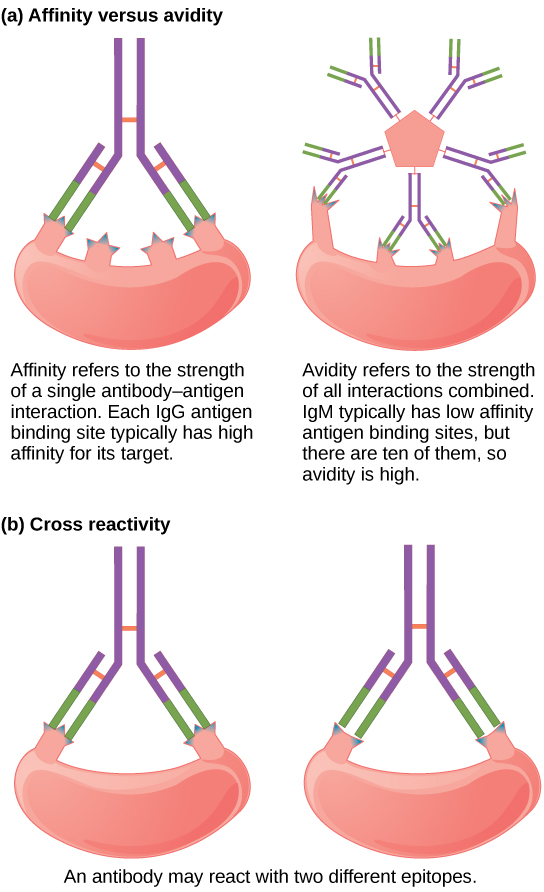
Affinity is the force of binding of one antibody molecule’s paratope with its corresponding epitope on the antigen molecule. It describes the interaction in an individual binding site. Knowledge of the affinity as well as construct and antigen properties is essential for evaluating the link between affinity and potency for a functional antibody. It is based on the same thermodynamic principles which govern any reversible biomolecular interaction, the same binding strength that enable your immune systems to fight off infections. And the strength refers to the strength of a specific bond between an antibody and its antigen. Precise and accurate evaluation of the affinity for an antigen-antibody interaction is an important measurement to establish affinity limits and the potency ceiling for a therapeutic antibody. In thermodynamic terms, affinity is expressed as the summation of all attractive forces resulting in increased binding strength and repulsive forces resulting in decreased binding strength.
What is Avidity?
Avidity is yet another binding concept which is often confused with affinity; in fact, both the terms refer to the concept of binding strength, except avidity refers to the total strength of the antigen-binding capacity of the mixture of polyclonal antibodies. Avidity comes to picture when a multivalent antigen binds to a bivalent antibody in solution or when an antibody binds to two monovalent antigen molecules on a surface. Unlike affinity, avidity describes the multivalent binding between multiple binding sites of the ligand and ligate, respectively. In simple terms, avidity is the binding force between a multivalent antibody and a multivalent antigen. Avidity cannot be described by thermodynamic terms, and is obtained by kinetic measurements. The avidity of the antibody depends on the affinities of the individual antibody binding sites, but is greater than the binding affinity as all the antibody-antigen interactions must be broken simultaneously for the antibody to dissociate completely.
Difference between Affinity and Avidity
Definition of Affinity and Avidity
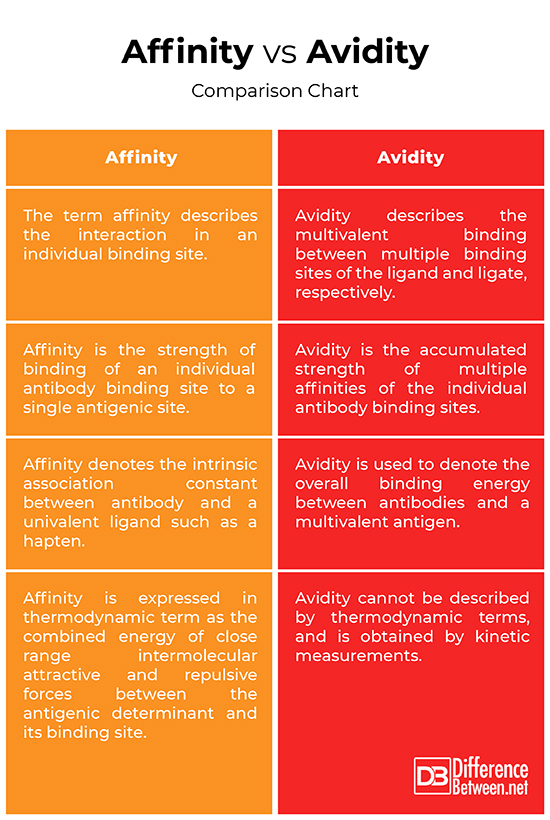
The terms affinity and avidity are sometimes inappropriately used as synonyms. In fact, both terms refer to the concept of binding strength. Affinity is defined in thermodynamic terms as the measurement of the degree of association between antibody and antigen, whereas the overall strength binding a multivalent antibody to a multivalent antigen is referred to as avidity, or otherwise known as “functional affinity.” Affinity is the strength of binding of an antibody combining site to one corresponding antigen epitope, whereas avidity is the total strength of the antigen-binding capacity of the mixture of polyclonal antibodies. Avidity is also referred to as functional affinity.
Occurrence
The term affinity denotes the intrinsic association constant between antibody and a univalent ligand such as a hapten, whereas the term avidity is used to denote the overall binding energy between antibodies and a multivalent antigen. The association constant is a measure of the affinity of the antibody for the epitope. Avidity cannot be described by thermodynamic terms, and is obtained by kinetic measurements. In thermodynamic terms, affinity is expressed as the summation of energy of close range intermolecular attractive and repulsive forces between the antigenic determinant and its binding site.
Biological Significance
Antibody affinity may play a critical role in determining biological activities of antibodies. So, it is necessary to be able to characterize an antibody response in terms of affinity as well as magnitude, and also to understand how antibody affinity may affect a particular assay. The term avidity is sometimes used synonymously with affinity but most often it is used to describe the ability of antibodies to form stable complexes with antigen. Avidity is the accumulated strength of multiple affinities of the individual antibody binding sites.
Affinity vs. Avidity: Comparison Chart
Summary of Affinity vs. Avidity
Despite their differences, the terms affinity and avidity are often indiscriminately used. Affinity is the force of binding of one antibody molecule’s paratope with its corresponding epitope on the antigen molecule and can be determined only by a single monovalent Fab fragment. It is the summation of all attractive forces resulting in increased binding strength and repulsive forces resulting in decreased binding strength. Avidity, on the other hand, is the binding force between a multivalent antibody and multivalent antigen, and is also referred to as functional affinity. The measured binding energy between antibodies and their corresponding antigens reflects the avidity of antibodies. Unlike affinity, avidity cannot be described by thermodynamic terms, and is obtained by kinetic measurements.
- Difference Between Caucus and Primary - June 18, 2024
- Difference Between PPO and POS - May 30, 2024
- Difference Between RFID and NFC - May 28, 2024
Search DifferenceBetween.net :
Leave a Response
References :
[0]Image credit: https://upload.wikimedia.org/wikipedia/commons/c/c7/Figure_42_03_04.jpg
[1]King, David J. Applications And Engineering Of Monoclonal Antibodies. Boca Raton, Florida: CRC Press, 1998. Print
[2]Shoenfeld, Yehuda, et al. Autoantibodies. Amsterdam, Netherlands: Elsevier, 2011. Print
[3]Bailon, Pascal, et al. Affinity Chromatography: Methods and Protocols. Berlin, Germany: Springer, 2000. Print
[4]Deshpande, S.S. Enzyme Immunoassays: From Concept to Product Development. Berlin, Germany: Springer, 1996. Print
[5]Reddehase, Matthias J. Cytomegaloviruses. Poole, United Kingdom: Caister Academic Press, 2013. Print