Difference Between Glucose and Fructose
 Glucose vs Fructose
Glucose vs Fructose
While not everyone would classify themselves as ‘sweet tooth’, there are few people who would gladly give up all sugar from their diet. Sugar can take many forms but the most common are sucrose, glucose, and fructose. If one is searching for the lowest common denominator, there should then just be glucose and fructose because these two monosaccharides are the building blocks of sucrose.
There are many similarities between glucose and fructose. They are both simple sugars, and are monosaccharides. Simple sugars contain only one type of carbohydrate as opposed to two like the disaccharide sucrose. The chemical formula for glucose and fructose are also the same: C6(H2O)6. Once they have entered the body, both sugars eventually make their way to the liver to be metabolized. Most processed and natural foods out there contain a combination of fructose and glucose. Even foods that you would expect to be nearly all fructose, such as high fructose corn syrup, actually have a 55%-45% composition in favor of fructose.
There are a few key ways in which these two sugars differ though.
Molecular Composition
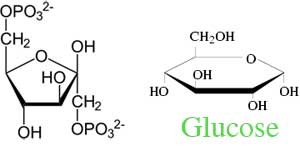
While their chemical formula is the same, the molecules of glucose and fructose are laid out in different formations. They both start out by making a hexagon with their six carbon atoms. Each carbon is bound to a water molecule.
Glucose ‘“ is an aldohexose. Its carbon is attached to a hydrogen atom by a single bond and an oxygen atom by a double bond.
Fructose ‘“ is a ketohexose. Its carbon is attached only to an oxygen atom by a single bond.
Metabolism
As aforementioned, both sugars end up in the liver. However,
Glucose ‘“ is eaten, absorbed into the blood stream, and makes it way to the liver where it is broken down to supply energy to the entire body. This breaking down process requires insulin.
Fructose ‘“ is eaten and absorbed but releases its energy slower than glucose. It does not need insulin to be metabolized and therefore is a marginally better choice for diabetics.
Taste
Fructose is many times sweeter than glucose. Many people feel that uncooked fructose can actually be overwhelming. This is especially true when the fruit that fructose is mostly found in becomes overripe. Once fructose has been cooked though, it loses much of its sweetness. That is why sucrose, or granulated sugar, is recommended for baking instead of crystallized fructose.
Summary
1.Fructose and glucose are both monosaccharides with the same chemical composition but a different molecular structure.
2.These two sugars are found in some combination in nearly all sweetened foods available.
3.Glucose needs insulin for proper metabolizing while fructose needs no insulin to be processed.
4.Raw fructose is many times sweeter than glucose.
- Difference Between Schizophrenia and Psychosis - March 7, 2024
- Difference Between African and Asian Elephants - March 7, 2024
- Difference Between Sunscreen and Sunblock - February 15, 2024

My husband has high uric acid. He also has Barres Epstein Virus. When he eats a lot of sugar his
blood ph balance gets out of whack. Would it be better for my husband to use raw unprocessed
Aguava Nectar for a sugar substitute, since it is mostly natural fructose & doesn’t pass into his blood-
stream? Is it true that natural fructose will not increase his uric acid levels?
Watch “Sugar: The Bitter Truth” on youtube.
he needs cell u loss
thank you for your information about glucose and fructose.
Thanks for the info, although part of it is not exactly right. Your body doesn’t need insulin to break down glucose, it needs insulin for glucose to get from the blood stream into the cells to be metabolized. Insulin is like a key, that unlocks cells so that glucose can go in- it has nothing to do with the breakdown process of glucose.
please
sent me first year biology notes [complete and easy]
Nice piece