Differences Between Glucose and Sucrose
Glucose vs. Sucrose
When one hears the terms glucose and sucrose, one automatically thinks of sugar and chemistry. These terms are common enough among chemists, food analysts, and those who check the nutritional content of chocolate or any other sugar-rich processed food. Chocolate and candy lovers are most likely familiar with sucrose, which is a common component of all sweets sold in the market. However, not everyone knows the differences between these two terms. Some people simply assume that glucose and sucrose are two sides of the same coin, as they both pertain to sugar. As a result, glucose and sucrose are often interchanged, and are treated as synonyms of sugar. When one gets to know these two terms better, however, the differences start to roll. Glucose and sucrose are not interchangeable terms. The only way to define and differentiate between these two terms would be via their chemical makeup.
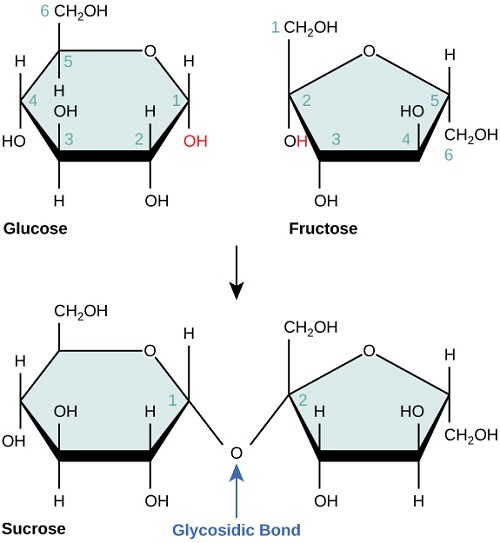
In formal chemistry terms, glucose is a monosaccharide known as C6H12O6 or C6(H2O)6. Glucose is a compound in the carbon group, and is considered as a hydrate, hence the term carbohydrate. Glucose has two forms, namely alpha and beta. On the other hand, sucrose is a disaccharide, a combination of fructose and glucose. Its formal chemical formula is known as C12(H2O)11. Sucrose is formed when the alpha form of glucose is mixed with fructose, which results in the loss of water and the formation of a disaccharide.
Because of its nature as a monosaccharide, glucose is easier for the body to break down and process as compared to sucrose. Sucrose, a disaccharide, is digested at a slower rate because it has a more complex chemical form. Glucose, a simple sugar, is broken down and absorbed more efficiently than sucrose. Because of this trend, the body first reduces disaccharides such as sucrose and other complex carbohydrates into glucose for efficient digestion.
The body utilizes glucose for energy. Absorption of glucose also signals the body to produce more leptin, an active hormone that deals with appetite moderation and storage of fat. In contrast, another hormone known as ghrelin is decreased in order to lessen the feeling of hunger. This is the reason why people who do not want to eat a lot, or want to cancel out the feeling of hunger, often resort to consuming a bar of chocolate or candy. The sucrose content of such food is high, which aids in eliminating hunger effectively.
If both terms were to be defined as sugar, glucose should be termed as simple sugar, while sucrose should be called table sugar. It should be noted that too much sugar consumption leads to serious health issues. A high intake of sugar, whether simple or table, leads to decreased production of insulin, increased liver fat, more bad cholesterol, and more visceral fat. Like any other food, sugar should be consumed moderately. Too much sugar can be detrimental to one’s health.
Summary:
1. The terms glucose and sucrose usually pertain to sugar. Sucrose is found in sweet food such as chocolate and candies.
2. Contrary to popular belief, glucose and sucrose cannot be used interchangeably. They have clear-cut differences, especially in their chemical composition.
3. Glucose is formally known as C6H12O6 or C6(H2O)6. It is a compound in the carbon group, and is considered a hydrate – hence the term carbohydrate. Glucose has two forms, namely alpha and beta.
4. On the other hand, sucrose is a disaccharide, a combination of fructose and glucose. Its formal chemical formula is known as C12(H2O)11. Sucrose is formed when the alpha form of glucose is mixed with fructose, which results in the loss of water and the formation of a disaccharide.
5. In layman’s terms, glucose should be correctly termed as simple sugar, while sucrose should be referred to as table sugar.
6. Consuming too much sugar can be detrimental to one’s health.
- Differences Between Fraternity And Sorority - January 8, 2014
- Differences Between Lucite and Plastic - January 7, 2014
- Differences Between Oil and Butter - January 6, 2014
Search DifferenceBetween.net :
Leave a Response
References :
[0]https://bio.libretexts.org/TextMaps/Map%3A_General_Biology_(OpenStax)/1%3A_The_Chemistry_of_Life/3%3A_Biological_Macromolecules/3.2%3A_Carbohydrates
